 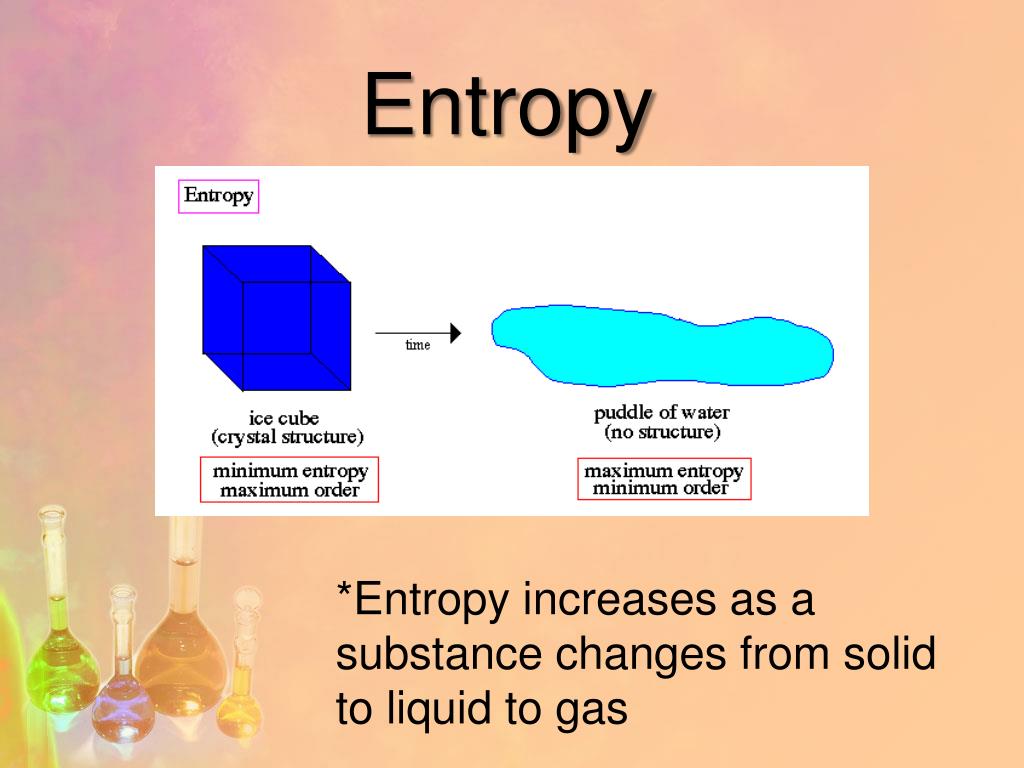
For example, after a cube of sugar has dissolved in a glass of water so that the sucrose molecules are uniformly dispersed in a dilute solution, they never spontaneously come back together in solution to form a sugar cube. Before discussing how to do so, however, we must understand the difference between a reversible process and an irreversible one. If you look at the Universe today and compare it to the Universe at any earlier point in time, you’ll find that the entropy has always risen and continues to rise, with no exceptions, throughout. (8) From this a possible decrease of entropy is calculated in an internal condensed process. Changes in entropy ( S ), together with changes in enthalpy ( H ), enable us to predict in which direction a chemical or physical change will occur spontaneously. In this case, the entropy with time would be defined as 10 r S(t)kPr(t)lnPr(t). 
For a full video: see Thus enthalpy is not the only factor that determines whether a process is spontaneous. 3.Decrease of Entropy and Condensed Process We discussed fluctuations magnified due to internal interactions, etc., so that the equal- probability does not hold. When water is placed on a block of wood under the flask, the highly endothermic reaction that takes place in the flask freezes water that has been placed under the beaker, so the flask becomes frozen to the wood. The reaction of barium hydroxide with ammonium thiocyanate is spontaneous but highly endothermic, so water, one product of the reaction, quickly freezes into slush. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
